| Our group has two research interests: (1) Novel Synthesis and Assembly Strategies as well as Thier Properties of Organic-inorganic Nanocomposite Colloildal Spheres; (2) Synthesis of Resins and Functional Coatings (high-temperature resistant, corrosion resistant, antifouling, thermal insulating, color changeable, self-cleaning and etc.). Typical results are briefly described as follows:
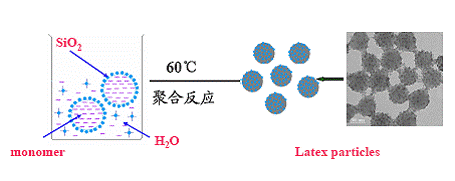
1. New Synthesis Strategies of Colloidal Spheres Organic-inorganic nanocomposite spheres can not only avoid the aggregation of nanoparticles and decrease the cost, but also produce novel mechnical, chemical, electric, magnetical and optical performances, which will find a lot potential applications in composite materials, coatings, electronic/photonic devices, catalysts and so on. We have developed a series of novel assembly methods of organic-inorganic nanocomposite spheres via non-covalent bonds (acid-base, electrostatic, hydrogen-bonding and etc.), and prepared various organic-inorganic nanocomposite spheres with core-shell or raspberrylike structure. Many papers have been published on international famous journals such as Chem. Mater., Macromolecules, J. Phys. Chem., Langmuir and acquired very good comments from international colleagues. Our paper published in Langmuir, 2005, 21, 2124 has been cited over 100 times.
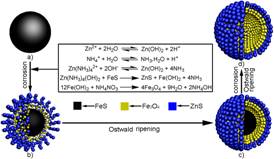
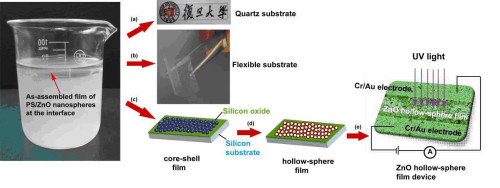
During the precess of our preparing organic-inorganic nanocomposite spheres, we found that polymer template particles could "dissolve" in basical alcohol/water medium, and invented a "one-step" strategy for fabrication inorganic hollow spheres. A series of monodisperse hollow inorganic spheres such as SiO2, TiO2, CaS, ZnO, Ag, Fe3O4/ZnS, Ta3N5 have been prepared. Our paper published in Adv. Mater. has been cited over 100 times; Our paper published in Adv. Funct. Mater. was used as the cover picture; We were invited to publish a critical review in Chem. Soc. Rev.
Representative papers: Chem. Soc. Rev.,2011, 40, 5472; 2012, 41, 4356; Adv. Mater., 2006, 18, 801; 2013, 25, 5343; J. Am. Chem. Soc., 2009, 131, 11276; ACS Nano, 2015, 9, 12513; Adv. Funct. Mater., 2007, 17, 1790; 2015, 25, 1035; Chem. Mater., 2005, 17, 3587; 2008, 20, 3251; Chem Comm., 2013, 49, 7225; 2014, 50, 5767; J. Mater. Chem. 2011, 21, 11276; 2011, 21, 17087; 2012, 22, 11245; J. Mater. Chem.-A, 2013, 1, 2183; 2014, 2, 14004; 2014, 2, 19771; J. Mater. Chem-C., 2015, 3, 10907; Macromolecules, 2004, 37, 9613; 2005, 38, 6411; 2014, 47, 1914; J. Phys. Chem. B, 2006, 110, 388; 2008, 112, 16; 2008, 112, 6536;J. Phys. Chem. C, 2007, 111, 11829; 2007, 111, 11692; Langmuir, 2005, 21, 2124; 2006, 22, 3858; 2006, 22, 4674; 2006, 22, 10175; 2006, 22, 3858; 2006, 22, 6403; 2009, 25, 7646; 2009, 25, 3467; 2010, 26, 6115; 2010, 26, 11391; 2010, 26, 14271; 2014, 30, 12011; Polym. Chem. 2013, 4, 3020; 2013, 4, 3293; 2015, 6, 3913
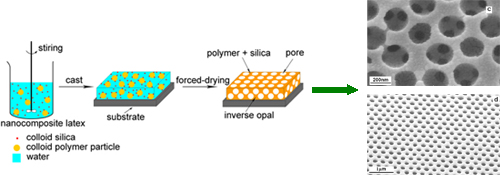
2. Self-assembly Strategies and Properties of Colloidal Spheres Recently, we report for the first time that "soft" polymer latex can form different structure and morphology at various film-forming temperatures be the aid of colloidal silica particles. For, example, when forced-drying at high temperature, it can directly form 2D or 3D ordered porous structure (inverse opal structure); When dried at room temperature, three-dimensional colloidal crystal films with large-area and robust mechanical properties can be successfully fabricated. Compared to the traditional self-assembly strategy of colloidal spheres, these approaches are very simple, the colloidal crystal films were easily obtained by casting, brush coating, spin coating or spray coating. Moreover, it can not only produce robust crystal films with enough mechanical properties to be further processed, but also be easily used for mass of production of crystal films with any sizes from square centimeters to square meters. These works have been published on Chem. Mater., J. Mater. Chem., Macromolecules, J. Phys. Chem. etc.
Representative papers: Chem. Soc. Rev., 2012, 41, 1350; Adv. Mater., 2011, 23, 1988; 2011, 23, 585; 2012, 24, 5872; Adv. Funct. Mater., 2012, 22, 998; 2012, 22, 1229; 2014, 24, 934; 2015, 25, 3256; Adv. Energy Mater., 2013, 3, 1636; Small., 2011, 7, 2449; 2014, 10, 3038; Chem. Mater., 2010, 22, 3433; 2012, 24, 3800; J. Mater. Chem., 2009, 19, 3594; 2011, 21, 687; 2012, 22, 944; 2012, 22, 17671; 2012, 22, 25207; 2012, 22, 8069; J. Mater. Chem.A, 2013, 1, 3146; 2013, 1, 10646; Macromolecules, 2008, 41, 6624; 2009, 42, 3591; 2013, 46, 7018; J. Phys. Chem. B, 2008, 112, 7706; Langmuir, 2010, 26, 6604; 2013, 29, 13503; Scientific Reports 2014, 4, 6847; 2015, 4, 12100
3. Research and Development of Functional Coatings We have done in-depth and systematic research on nanocomposite and organic-inorganic nanohybrid coatings very early in the world, including embedding ZnO, Al2O3, SiO2, TiO2, ZrO nanoparticles or their networks as well as POSS into water-borne, solvent-based or UV-curable resins and coatings via blending method or in situ polymerization, understanding the relationship between preparation-structure-property of organic-inorganic nanohybrid coatings and developing a series of functional coatings such as self-cleaning, high-solid, mar-resistant and termal-insulating coatings. These works have been paid attention from academa and companies: more than 70 papers have been published on international famous journals such as J. Mater. Chem., Polymer, Prog. Org. Coat., J. Colloid Interface Sci.; Over 27 China patents plus 1 US patent have been approved. Quite several projects have been awarded by central and local governments. Many vice-presidents or technique directors from international famous chemical companies including GE, Dupont, Dow Chemical, ICI, GE, Bayer, Henkel, Merck, BASF, PPG, BEHR, Degussa, Huntsman, Siegwerk have visited our lab to seek cooperation. Honored by People’s Republic of 2009 Sci. & Tech. Invention Silver Award in 2 5752; Prog. Org. Coat., 2002, 45, 33; 2014, 77,1015; 2010, 67, 302; 2015, 85, 208., 2014 1, 1547; 2013,, 52, 122; J. Mater. Chem. C,2011, 3, 19093; Polymer, 2015, 21, 6161; J. Mater. Chem-A, 2011, 50, 3609; J. Mater. Chem., 2009, 47, 1640; Polymer, 2006, 45, 8127; Polymer, 2004, 45, 2967; Polymer, 2004Polymer, , 50, 11891;2014ChemCommuns, 1035; 25, ,2015 10, 1386; Adv. Funct. Mater., 2016, ACS Nano., Representative papers: |
Field
Copyright©2019 Fudan
Add: Shanghai Yangpu Fudan University Tel.: (86)021-55664033
Support By:Weicheng